Does wind have weight? The truth about air, atmospheric pressure, and why you're not crushed
Discover if wind actually has weight and understand how billions of tons of air above you don't crush you. The answer involves atmospheric pressure, air density, and surprising physics.
Does wind have weight? The truth about air, atmospheric pressure, and why you’re not crushed
Yes, wind has weight — because wind is air in motion, and air has mass. And if air has mass, gravity pulls it down, which means it has weight. In fact, the entire atmosphere weighs about 5.15 × 10¹⁸ kg (that’s a 5 followed by 18 zeros). The real question isn’t whether air weighs anything, but why the hell don’t we feel this weight crushing us.
When I first started studying atmospheric pressure, my brain glitched. Like, how is it possible to have a column of air kilometers high above my head and not feel anything? The answer is one of the most elegant things in physics — and it involves pressure, equilibrium, and the fact that our body is basically a pressure-compensation machine.
And that’s what I need to write about today. Because this seemingly simple question — “does wind have weight?” — opens the door to understanding how the atmosphere works, why planes fly, why your ears pop in the mountains, and even why you wouldn’t explode in space (spoiler: you would, but not the way movies show it).
Air weighs (and much more than you imagine)
Let’s start with the basics: air is matter. It’s made primarily of nitrogen (78%) and oxygen (21%), with bits of argon, CO₂, and other gases. And like all matter, air has mass.
The density of air at sea level is approximately 1.225 kg/m³. This means that each cubic meter of air — imagine a cube of 1m × 1m × 1m — weighs about 1.2 kg. Doesn’t sound like much? But now think about the absurd amount of air that exists above you.
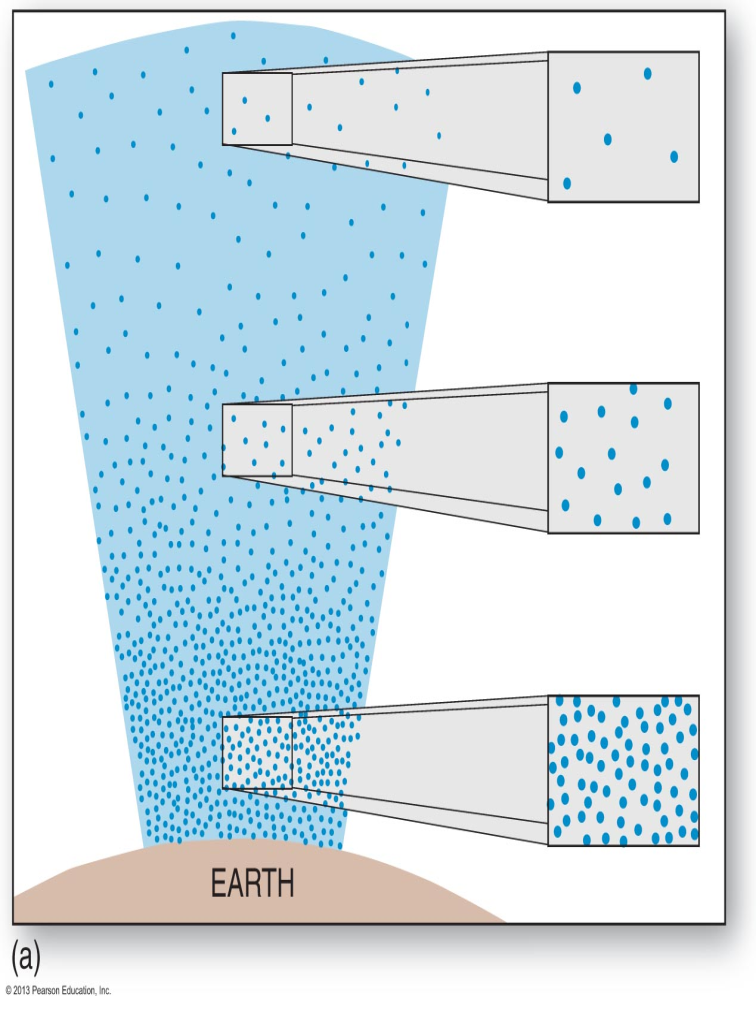
Earth’s atmosphere is about 100 km high (technically it goes beyond that, but it becomes so thin it’s irrelevant). If you take that entire column of air above your head and calculate the weight, you get something surprising: each square meter of Earth’s surface supports approximately 10,000 kg of air — that is, 10 tons.
Per square meter. Above your head. Right now.
I know. It’s unsettling when you stop to think about it.
Atmospheric pressure: the weight of air pushing everything
This weight of air creates what we call atmospheric pressure. Pressure is force per area — and in the case of the atmosphere, it’s the weight of that entire column of air divided by the area it covers.
At sea level, atmospheric pressure is approximately 101,325 Pa (Pascals) — or 1 atm (atmosphere), as it’s more commonly called. In more intuitive units, that’s about 1 kg of force per square centimeter (1 kgf/cm²).
Translation: every square centimeter of your body is being pushed by 1 kg of force coming from the surrounding air. If you have a body surface area of 2 m² (average adult), we’re talking about approximately 20,000 kg of force being applied to your body by the atmosphere.
Twenty tons. Crushing you. All the time.
But wait — if there are 20 tons of pressure trying to crush you, why don’t you turn into human purée?
Why you’re not crushed: the equilibrium trick
Here’s the kicker: atmospheric pressure acts equally in all directions.
It’s not just from top to bottom. It’s from bottom to top, from the sides, from inside to outside. The atmosphere pushes you with the same force in all directions at the same time — which means the forces cancel out.
But there’s more: the air inside your body (in the lungs, sinuses, stomach, ears) is also at the same pressure as the external air. This creates an equilibrium. Internal pressure compensates for external pressure. It’s like being at the bottom of a pool — water pushes you from all sides, but you’re not crushed because your body is made of liquids and tissues that are at the same pressure.
That’s why rapid pressure changes are so problematic. When you climb a mountain too fast, external pressure decreases, but internal pressure (in your ears, for example) is still high — and you feel that “clogged ear” sensation. Your ears need to equalize the pressure, usually by popping or opening the Eustachian tube.
And that’s why, if you went to space without a spacesuit, you wouldn’t explode like a balloon — but the pressure difference would cause other serious problems (bodily fluids boiling, lungs collapsing, etc.). But that’s talk for another post.
And wind in all this?
Now that we know air has weight and creates pressure, the question about wind becomes clearer: wind is just air moving from an area of high pressure to one of low pressure.
When air heats up (from the Sun, for example), it becomes less dense and rises — creating a low-pressure area. The cooler, denser air from the surroundings moves to fill this space — and boom, you have wind.
So yes, wind has weight. A strong gale can push you with considerable force — and it’s no wonder. Fast-moving air can exert a very high dynamic pressure. It’s the same principle that makes airplanes fly: fast air on top of the wing creates low pressure, slower air underneath creates high pressure, and the difference generates lift.
Speed matters. A 100 km/h wind has much more “effective weight” pushing you than a 10 km/h breeze — because wind force increases with the square of velocity. Double the speed, quadruple the force. That’s why hurricanes are so destructive.
Density changes everything: mountain vs. sea level
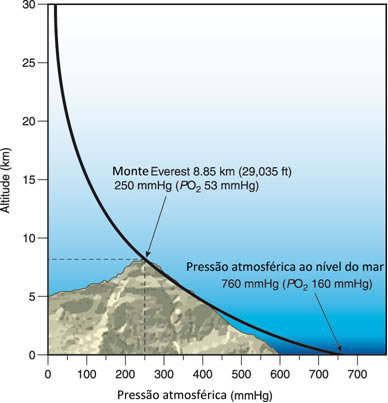
Here’s a cool thing: atmospheric pressure decreases with altitude. At the top of Mount Everest (8,848 m), the pressure is only about ⅓ of the pressure at sea level.
Why? Because there’s less air above you. Remember that 100 km column of air? If you’ve already climbed 8.8 km, only 91.2 km remain — and air density decreases exponentially as you go up (most of the atmospheric mass is concentrated in the first 10-15 km).
That’s why climbers on Everest need supplemental oxygen. It’s not that the air is “different” — it’s still ~21% oxygen. But with ⅓ of the pressure, each breath brings ⅓ of the amount of oxygen molecules. Your body needs to compensate by breathing faster, and even then it may not be enough.
And here’s a detail I love: air density also changes with temperature. Hot air is less dense (molecules spread apart), cold air is denser (molecules come closer). That’s why hot air balloons rise — the air inside the balloon is less dense than the surrounding air, creating buoyancy.
Measuring pressure: why the barometer exists
The first scientists to understand atmospheric pressure were geniuses. Evangelista Torricelli, in 1643, created the first barometer — a glass tube filled with mercury, inverted in a basin. The mercury descended a bit, but stopped at about 760 mm height.
Why? Because the pressure of air pushing the mercury in the basin was enough to support that 760 mm column. If atmospheric pressure increased (good weather coming), the mercury rose. If it decreased (storm coming), the mercury fell.
To this day, one of the pressure units is mmHg (millimeters of mercury) — also called Torr, in honor of Torricelli. Normal atmospheric pressure = 760 mmHg = 1 atm = 101,325 Pa.
Questions I had (and the answers)
“If air has weight, why do helium balloons rise?”
Because helium is less dense than air. The buoyancy (upward force that air exerts) is greater than the weight of balloon + helium, so it rises. It’s the same principle as floating in water — wood floats because it’s less dense than water.
“Can you ‘feel’ the weight of air?”
Not directly, because we’re in equilibrium with it. But you feel pressure changes — when you climb a mountain, dive underwater, or fly in a plane. Ears “popping” is your body trying to equalize internal pressure with external pressure.
“If the atmosphere weighs so much, why doesn’t it ‘fall’ to the ground?”
It is on the ground — gravity pulls air down, creating atmospheric pressure. What keeps the atmosphere “in place” is the balance between gravity (pulling down) and pressure (pushing up from lower layers). It’s the same reason the ocean doesn’t “fall” to the bottom: the layers on top push the ones below.
“Why does pressure decrease with altitude if gravity is still pulling?”
Because there’s less air mass above you. Gravity is still the same, but you have less “air column” pressing on you. Each air molecule still weighs, but there are fewer of them stacked on top.
“Does vacuum have weight?”
No. Vacuum is absence of matter — so it has no mass, therefore no weight. But creating vacuum on Earth is difficult, because atmospheric pressure tries to “crush” any empty space. That’s why cans crumple when you remove the air inside them.
Final thoughts (and why this fascinates me)
I find it incredible that we spend our entire lives swimming in an ocean of air and rarely stop to think about it. There are literally tons of mass above your head right now, pressing you with absurd force, and you don’t even notice. It’s the kind of thing that, when you really understand it, changes how you see the world.
Like, wind isn’t just “air moving.” It’s mass in motion, transferring energy, creating force. It’s the planet redistributing heat, balancing pressures, sustaining life. Every gust you feel is a little piece of physics in action — dense air rushing to replace less dense air, equalize temperature, balance the system.
And atmospheric pressure? It’s the reason you can breathe. It’s the reason liquids don’t boil at room temperature (well, actually they do, but only at low pressure — try boiling water on Everest and it boils at about 70°C). It’s the reason planes fly, submarines sink, and balloons rise.
When I stop to think that we’re living at the bottom of an ocean of air, and that this ocean protects us from cosmic radiation, keeps temperature stable, distributes water across the planet, and still gives us something to breathe… it’s hard not to be fascinated.
And there’s something poetic in all this: you’re made of the same stuff as the entire universe — atoms that came from stars that exploded billions of years ago. And these atoms, organized in the shape of “you,” are in perfect equilibrium with a 100 km column of other atoms above your head. You’re part of the system. You are the system.
💡 Summary in 3 points:
- Wind has weight because air has mass — the entire atmosphere weighs 5.15 × 10¹⁸ kg, creating about 10 tons of pressure per square meter at sea level.
- You’re not crushed because pressure acts equally in all directions — and your body is in internal/external equilibrium, compensating the forces.
- Atmospheric pressure decreases with altitude — less air above you = less weight = less pressure. That’s why Everest is so hard to climb (⅓ of normal pressure).
References:
-
NASA Earth Observatory: Atmospheric Pressure earthobservatory.nasa.gov
-
National Weather Service: Air Pressure and Weather weather.gov
- HEWITT, Paul G. Conceptual Physics. 12th ed. Pearson, 2014.
-
NOAA: Understanding Air Pressure noaa.gov
Personal note: I got curious about how atmospheric pressure affects biological processes — like, how do plants regulate transpiration based on pressure? And how have altitude animals (like condors) adapted to live with less available oxygen? For another post.